A Langmuir film can be defined as an insoluble monolayer of atoms, molecules, or even nanoparticles floating at the liquid-gas interface (or liquid-liquid). Monolayer formation is possible due to the forces of self-assembly on insoluble molecules at the surface of a liquid.
Many materials can form Langmuir films
When molecules possess hydrophobic (water-hating) and hydrophilic (water-loving) parts, they may orientate themselves in a predictable way on the water surface, across the entire film, opening up amazing possibilities for further controlling the architecture of deposited molecular films. Mixtures of molecular materials may be combined to create entirely new properties within a monolayer. These properties may be tunable through control of the packing density, thereby controlling the extent of neighbor-neighbor molecular interactions.
Many molecular materials are well suited to forming insoluble layers at the air-water interface. These include lipids, nanoparticles, polymers, proteins, and many other biomolecules. Modern chemical engineering has made it possible to synthesize almost any type of functional molecule with hydrophobic appendages that make them insoluble in water and suitable for creating monolayers. Recent research has shown novel compounds such as carbon nanotubes, nanowires, or graphene form Langmuir films.
Many of these amphiphilic substances that are insoluble in water can, with the help of a volatile and water-insoluble solvent, easily spread on the water to form an insoluble monolayer at the air/water interface. These monolayers or Langmuir (L) films represent the most extreme case when considering adsorption to interfaces since all molecules are concentrated in a single-molecule thin layer. The amphiphilic nature of the molecules dictates the orientation at the interface (air/water or oil/water) in such a way that the polar head group is immersed in the water and the long hydrocarbon chain points towards the air, gas or oil. The name Langmuir film originates from the name of the pioneer of these films Irving Langmuir.
Hydrocarbon chains on substances used in monolayer studies must be long enough to form an insoluble monolayer.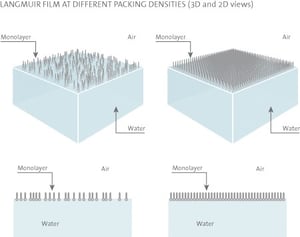 A rule of thumb is that there should be more than 12 hydrocarbons or groups in the chain ((CH2)n, n > 12). If the chain is shorter, though still insoluble in water, the amphiphile on the water surface tends to form micelles. These micelles are water-soluble, which prevents the build-up of a monolayer at the interface. On the other hand, if the length of the chain is too long the amphiphile tends to crystallize on the water surface and consequently does not form a monolayer. It is difficult to determine the optimal length for the hydrocarbon chain because its film-forming ability also depends on the polar part of the amphiphile. Furthermore, the amphiphile has to be soluble in a highly volatile organic solvent which is water-insoluble (chloroform or hexane is commonly used).
A rule of thumb is that there should be more than 12 hydrocarbons or groups in the chain ((CH2)n, n > 12). If the chain is shorter, though still insoluble in water, the amphiphile on the water surface tends to form micelles. These micelles are water-soluble, which prevents the build-up of a monolayer at the interface. On the other hand, if the length of the chain is too long the amphiphile tends to crystallize on the water surface and consequently does not form a monolayer. It is difficult to determine the optimal length for the hydrocarbon chain because its film-forming ability also depends on the polar part of the amphiphile. Furthermore, the amphiphile has to be soluble in a highly volatile organic solvent which is water-insoluble (chloroform or hexane is commonly used).
Control Langmuir film packing density
The free movement of molecules at the interface of a Langmuir film provides great flexibility for the control of the packing density and the study of monolayer behavior. Once compressed, a monolayer film can be considered to be a two-dimensional solid film with a surface area-to-volume ratio far above that of bulk materials. Under these conditions, materials often yield fascinating new properties. A Langmuir Trough allows you to infer how particular molecules pack together while confined in two dimensions (a set of barriers is used to compress the monolayer at the interface). The surface pressure-area isotherm can also provide a measure of the average area per molecule and the compressibility of the monolayer.
In a typical isotherm measurement, a monolayer is organized under compression, starting from a two-dimensional gas phase (G) moving through a liquid phase (L) to a fully organized solid phase (S). 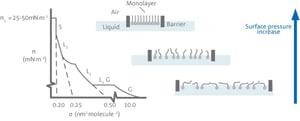 In the gas phase, the molecules are not interacting with each other. When the surface area is decreased the molecules become more closely packed and start to interact with each other. In the solid phase, the molecules are completely organized and the surface pressure increases dramatically. At the maximum surface pressure, the collapse point is reached after which the monolayer packing is no longer controlled. Under certain conditions, Langmuir films can be transferred to solid surfaces for further studies or simple coating.
In the gas phase, the molecules are not interacting with each other. When the surface area is decreased the molecules become more closely packed and start to interact with each other. In the solid phase, the molecules are completely organized and the surface pressure increases dramatically. At the maximum surface pressure, the collapse point is reached after which the monolayer packing is no longer controlled. Under certain conditions, Langmuir films can be transferred to solid surfaces for further studies or simple coating.
