Proteins are vital for life and perform a wide range of essential biochemical tasks in all living organisms. Cells of these organisms are hence under a constant pressure to maintain an optimal protein environment, assuring all proteins are correctly folded and functional.
Unfolded proteins are sticky and tend to form so-called protein aggregates with either themselves, other proteins or when binding to exposed surfaces within the cell. Aggregation mechanisms depend on both primary amino acid sequence of the protein and external environment such as pH, salt and temperature. Most protein aggregates can be reversed or degraded by the cell protein quality system (molecular chaperones or proteases).
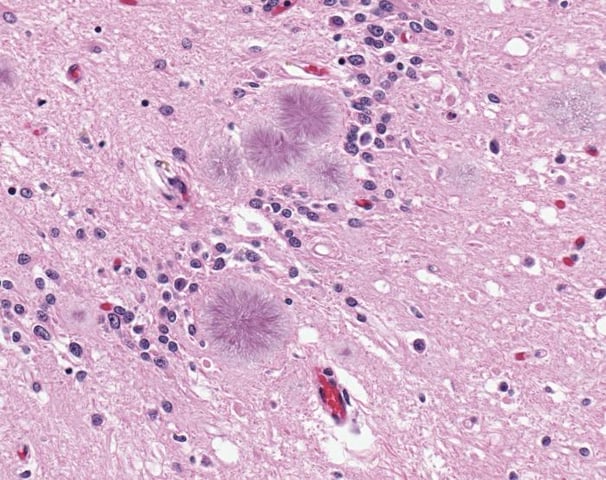
However, sometimes the control mechanisms fail and accumulated aggregates transform into amyloid plaques and other protein megastructures. This is the case in protein misfolding diseases, proteopathies, such as Alzheimer’s and Parkinson’s disease. Amyloids behave very differently to functional soluble protein, for example in regards to rigidity. Current trends within this field of research is to study the kinetic buildup of such megastructures and consequently also searching for therapeutic agents that prevent buildup and hence disease. Protein misfolding diseases are a huge threat to the increasing ageing population, with such diseases affecting more than 10% of all people over the age of 65.
Learn about how aggregation of protein Tau in tauopathies, a sub-set of neurodegenerative diseases, can be studied with QCM-D.
QSense QCM-D reveals lipid bilayer formation and hydrated model membrane structures that can be difficult to capture with other methods.
QSense QCM-D reveals in real time how thin films degrade and are removed, from cleaning and detergency to corrosion, etching and degradable coatings.
Learn how QSense QCM‑D reveals time‑resolved reagent–mineral interactions, linking surface chemistry to flotation performance in mineral processing.
Learn how QSense QCM-D monitors crosslinking and collapse of thin films by tracking hydrated mass and mechanichal properties
Learn how QSense QCM-D reveals protein–surface interactions and adds interface-focused insight to biopharmaceutical formulation and stability work
Learn how QSense sensors enable application‑relevant biointerface interaction analysis and explore our sensor offering for different areas
Learn how QSense QCM D can be used to analyze swelling of thin films, including magnitude and dynamics.
Read about how molecule-surface interaction processes such as adsorption and desorption can be analyzed with QCM-D.
Learn best practices and step-by-step methods for accurate QCM-D coating thickness measurement on QSense sensors using QSense Omni.
Compared to QCM, QCM-D measures an additional parameter, and provides more information about the system under study.
Discover how QCM-D analysis reveals real-time etching dynamics, helping optimize cleaning processes and protect surfaces from unwanted damage.
