
Single-sheet graphene (SG) is the first truly two-dimensional material and has been shown to have many outstanding material properties, such as high electrical and thermal conductivity and high tensile strength. The 2010 Nobel Prize in Physics was awarded to Professors Andre Geim and Konstantin Novoselov (Manchester University) for their ground-breaking research on the two-dimensional material graphene.
Many believe graphene is one of the most promising and versatile material discovered. Potential applications for the material include building smaller and faster electronic circuits, developing stronger and more flexible construction materials, and creating more efficient electric batteries, to name a few.
Graphene can act as both n- and p-type conductors and due to the semiconducting nature, this has raised some speculations about it replacing silicon in electronics in the future. Because of the electrical properties and transparency of SG layers combined with a good chemical resistance, one of the most interesting applications for graphene is for it to be used instead of indium tin oxide (ITO) or fluorine tin oxide (FTO) in optoelectronics such as solar cells and light emitting diodes.
Deposition of graphene has been one of the main factors hindering the use of graphene in industrial applications. Mechanical exfoliation has been the primary method to produce single-sheets of graphene. Although an experienced scientist can produce a single graphene sheet by simply peeling it with tape from a graphite flake, it is not a method that can be scaled to industrial use. Alternative methods for mechanical exfoliation are thus looked for.
For industrial applications, the deposition method must have;
• High quality in the 2D crystal lattice to ensure high mobility
• Fine control over the crystalline thickness
• Ease of integration
• Scalability
A possible solution is offered by graphene oxide. Graphene oxide is chemically modified graphene containing oxygen functional groups such as epoxides, alcohols, and carboxylic acid. 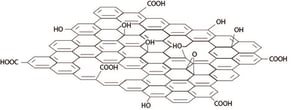 Graphene oxide has received a great deal of attention because it readily exfoliates as a single sheet in water. Although graphene oxide itself is not conductive, it can be reduced to graphene by thermal or chemical treatments after deposition. Most exiting advantages of the graphene oxide method are its low-cost and scalability. Some of the most used deposition methods for graphene oxide include spray, spin and dip coating and Langmuir - Blodgett and Langmuir-Schaefer deposition.
Graphene oxide has received a great deal of attention because it readily exfoliates as a single sheet in water. Although graphene oxide itself is not conductive, it can be reduced to graphene by thermal or chemical treatments after deposition. Most exiting advantages of the graphene oxide method are its low-cost and scalability. Some of the most used deposition methods for graphene oxide include spray, spin and dip coating and Langmuir - Blodgett and Langmuir-Schaefer deposition.
If you would like to read more about the deposition methods and their pros and cons, please download the short overview below.
Nanoparticle thin films are gaining recognition and use in various products and applications including displays, sensors, and energy storage.
LB films offer various opportunities for the creation of biosensors.
Roll-to-roll LB technology combines the benefits of Langmuir-Blodgett to high throughput and the possibility to use flexible substrates.
Spin coating is one of the most used thin-film coating technique and for this reason, any new technique is typically compared to it.
Nanoparticle thin films are finding applications in areas where functional coatings are needed. Dip coating offers one of the simplest methods to coat the substrate with nanoparticles.
This blog shows that nanoparticle deposition can improve thin film solar cell efficiency.
Describes the most common nanolithography methods to produce nanosized features
Nanoparticle lithography is a promising fabrication technique. Unlike other techniques, it has a potential to be performed on curved surfaces.
