Surface tension of inkjet inks and the wettability of the printing substrate are important factors influencing the final printing quality and process reliability. Surface tension and interfacial interactions can be explored with various tensiometry technologies: Equilibrium and dynamic surface tension measurements can be utilized in ink formulation and development.
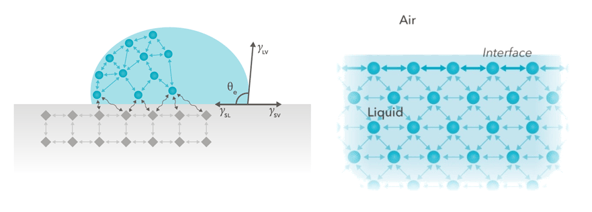 Figure 1. Forces at an interface originate from the fact that the molecules interact differently with surrounding molecules at the interface than those in the bulk.
Figure 1. Forces at an interface originate from the fact that the molecules interact differently with surrounding molecules at the interface than those in the bulk.
In addition to the sole surface tension measurements, also critical micelle concentration (CMC) can be used to define the point when surface tension becomes independed of surfactant concentration, and interfacial rheology experiments to provide information about interfacial stability. Since the printed image is created by the ink-droplets, the contact angle measurement is a useful tool to demonstrate the ink-substrate interactions. Contact angle measurement enables also definition of the printing substrate’s surface free energy.
Applying these techniques on real-life samples and processes means we have to take into account factors causing contact angle hysteresis and small size of the inkjet droplets. For example, it is well-known that both surface chemistry and roughness have an influence on wetting, and that this phenomenon can be understood further by correcting the contact angles for roughness.
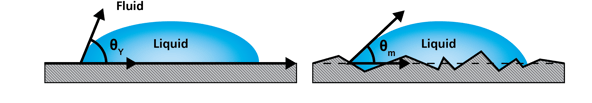 Figure 2. Influence of surface roughness on wetting can be taken into account in the wetting analyses.
Figure 2. Influence of surface roughness on wetting can be taken into account in the wetting analyses.
Droplet size influence
Since the inkjet inks droplets are very small, penetrative and low viscous, it sets a high demand on the surface properties of substrates. The contact angle analyses allow studying the droplet spreading and absorption via analyzing the drop dimensions and contact angles with time.
The typical droplet size used in contact angle measurements is between 1 and 10 microliters. It has been showed that droplet volume has no significant influence on the contact angle with close to-ideal surfaces, such as clean quartz plates. The ideal surface is considered to be smooth, rigid, chemically homogeneous, insoluble, and non-reactive. However, the larger a contact angle hysteresis is the greater impact droplet volume has on contact angle.
In recent years, the interest towards to the picoliter droplets has increased due to emerging inkjet technology and need to analyze small micropatterned and single fiber surfaces. Therefore, discussion about the effects of droplet size on contact angle has broadened to compare microliter droplets to picoliter droplets. The initial contact angle of picoliter droplet has been demonstrated to correlate best with the contact angle of microliter droplet. Due to fast evaporation of picoliter droplet, the contact angle decrease as a function of time reaching the receding angle.
 Figure 3. High speed camera enables to measure the initial pico¬liter drop contact angle right after the drop hits the surface and then the fast spreading and absorption phenomena.
Figure 3. High speed camera enables to measure the initial pico¬liter drop contact angle right after the drop hits the surface and then the fast spreading and absorption phenomena.
Learn more, watch this recorded webinar on Surface tension and wetting – Experimental approaches and significance in inkjet printing:
Learn about the effect of surface roughness and wettability on biocompatibility of biomaterials and medical devices.
This blog post describes the importance of fiber diameter on the contact angle measurements of fibers with Wilhelmy method
Standard contact angle measurement considers the surface's chemical properties. The influence of surface roughness is added by utilizing the Wenzel equation.
Wettability is crucial in biomedical applications as it affects protein adsorption, cell adhesion, blood coagulation, and bacterial colonization.
Liquids’ ability to wet a solid surface has widespread importance in many everyday products and industrial processes.
Membrane wettability is a key property to ensure success of membrane distillation process
Hydrophobic surface properties are needed in contact with the food product while hydrophilicity is needed when printing on food packaging.
Wettability is pivotal in pharmaceutical dosage form manufacturing as well as in drug efficacy.
Understanding the wettability of membranes is essential for optimizing these processes and achieving desired separation outcomes.
