One of the major challenges in pharmaceutical development is protein aggregation – a phenomenon that could have negative impact not only on the drug quality, but which could also potentially affect drug safety. In a recent study1, results are presented that provide a deeper understanding of the relation between protein adsorption and aggregation at the oil-water interface. The new insights could help designing more stable therapeutic formulations, and QSense QCM-D was one of the analysis methods that helped piecing the puzzle together.
As we talked about in a previous blogpost, a common way to store and administer therapeutic protein is via prefilled syringes - syringes where the interior surfaces typically are coated with a lubricant, silicone oil, to facilitate the use of the syringe. As the therapeutic protein stored in the syringe interacts with this hydrophobic surface it could adsorb, a process which has been a suspected source of aggregation.
To get a deeper understanding of protein interaction with silicone-oil coated surfaces, and how this is potentially related to protein aggregation, a group of researchers set out to analyze the surface activity of monoclonal antibodies (mAbs)1, a major class of protein-based therapeutics used for the treatment of several different diseases. In the study1, the researchers also analyzed if two, pharmaceutically relevant, and commonly used, surfactants would lower the adsorption and aggregation of the mAbs under investigation.

To paint the full picture, and to get a deeper understanding of the surface activity and the aggregation of the mAbs, several complementary analysis methods were used1. The surface mass adsorption of the two mAbs, the two surfactants and mAb + surfactant mixtures was analyzed by QCM-D, a surface sensitive technology that detects time-resolved mass changes at the nanoscale.
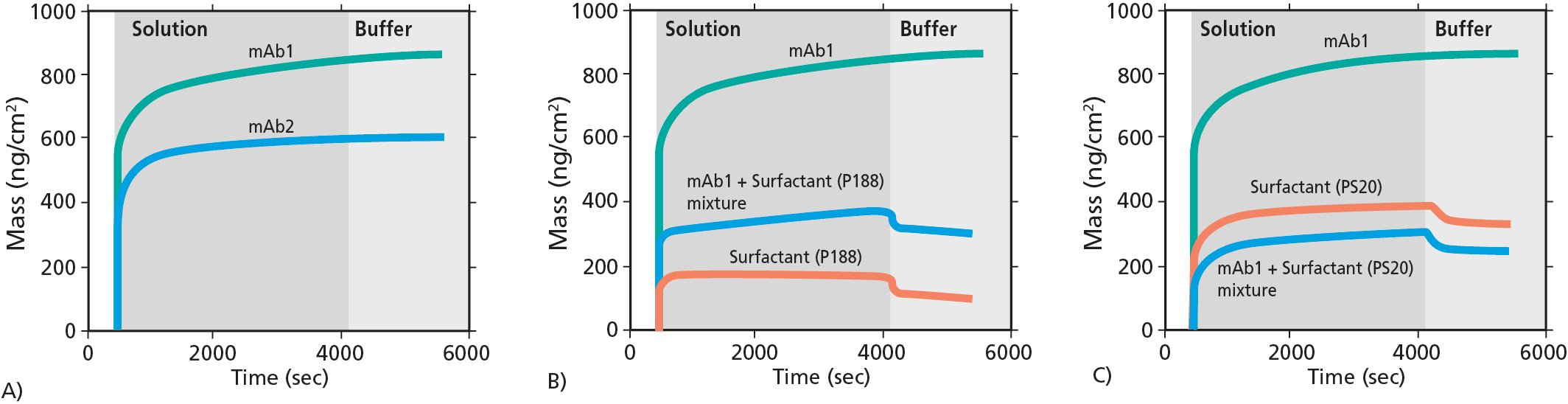
Figure 1. Mass uptake at the sensor surface measured by QCM-D. A) Comparison of mass uptake of mAb1 and mAb2. B) and C) comparison of mAb1, surfactants P188 and PS20, and mAb1 + surfactant mixtures.
The QCM-D analysis revealed several insights on the interfacial adsorption and the surface mass uptake of the mAbs, the surfactants and the mAb + surfactant mixtures1. For example, the QCM-D data, Fig 1., showed that
Combining the insights from the QCM-D analysis with information from the complementary characterization, the researchers concluded that there is a direct relation between mAbs adsorption at the oil-water interface and aggregation. The study also showed that surfactants, which will competitively adsorb to the interface, will lower the aggregation of the mAbs
Protein aggregation is one of the challenges encountered in pharmaceutical development. In a recent study1, results are presented that links absorption and aggregation, information that can help formulate more stable therapeutic solutions. In the study, the researchers used QSense QCM-D analysis to characterize the surface mass uptake - information which helped piecing the puzzle together.
Watch the webinar to learn more about this study1.
QSense QCM-D reveals lipid bilayer formation and hydrated model membrane structures that can be difficult to capture with other methods.
QSense QCM-D reveals in real time how thin films degrade and are removed, from cleaning and detergency to corrosion, etching and degradable coatings.
Learn how QSense QCM‑D reveals time‑resolved reagent–mineral interactions, linking surface chemistry to flotation performance in mineral processing.
Learn how QSense QCM-D monitors crosslinking and collapse of thin films by tracking hydrated mass and mechanichal properties
Learn how QSense QCM-D reveals protein–surface interactions and adds interface-focused insight to biopharmaceutical formulation and stability work
Learn how QSense sensors enable application‑relevant biointerface interaction analysis and explore our sensor offering for different areas
Learn how QSense QCM D can be used to analyze swelling of thin films, including magnitude and dynamics.
Read about how molecule-surface interaction processes such as adsorption and desorption can be analyzed with QCM-D.
Learn best practices and step-by-step methods for accurate QCM-D coating thickness measurement on QSense sensors using QSense Omni.
Compared to QCM, QCM-D measures an additional parameter, and provides more information about the system under study.
Discover how QCM-D analysis reveals real-time etching dynamics, helping optimize cleaning processes and protect surfaces from unwanted damage.
Discover how QSense QCM-D helps tackle fouling challenges across industries
