
Critical micelle concentration (CMC) is the surfactant concentration at which micelles start to form in solution. CMC is one of the key parameters in surfactant characterization because it shows how a system changes as concentration increases. Understanding CMC is important in applications ranging from detergency and emulsification to pharmaceuticals, cosmetics and advanced materials.
In this article, we explain what CMC means, why it matters in practice and how it is commonly measured, with focus on surface tension measurements.
To understand critical micelle concentration, or CMC for short, we need first to understand surfactants. Surfactants are amphiphilic molecules that have hydrophilic and hydrophobic parts (Figure 1A). When surfactants are added to water, they orient themselves at the air-water interface so that the hydrophilic part is in water and hydrophobic part in air (Figure 1B). Another energetically favorable structure is a micelle (Figure 1C), where hydrophobic parts are pointing inwards the spherical structure protected by the hydrophilic outer shell.
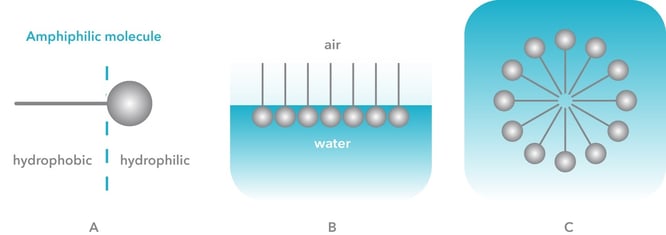
Figure 1. A) Amphiphilic surfactant molecule B) Surfactant molecules orient themselves at air-water interface C) At high concentrations, micelles are formed.
At low surfactant concentrations, molecules mainly adsorb at interfaces and reduce surface tension. As more surfactant is added, at some point the interface becomes saturated with surfactant molecules and micelles start to form in the bulk solution. The concentration at which micelles first form is called the critical micelle concentration (CMC). Above the CMC, most additional surfactant goes into micelles rather than further lowering surface tension, so CMC effectively defines how much surfactant is needed to reach the lowest useful surface tension for a given system.
It is important to remember that CMC is not a single universal number: the value depends on surfactant structure, temperature, ionic strength, pH, additives and on the method used for measurement. For that reason, CMC data should always be interpreted together with the conditions and technique used to obtain it.
The main reason to add surfactants to a solution is to decrease surface tension. When surfactants adsorb at interfaces, they make it easier for liquids to wet, spread, foam, emulsify, or solubilize other materials. CMC tells you how far you can lower the surface tension by adding surfactant. Above this point, further additions mainly increase micelle concentration rather than reduce surface tension further.
Knowing the CMC helps you understand how a surfactant system behaves as concentration changes. It is widely used if you want to:
Compare surfactants or formulations under defined conditions
For example, detergents typically contain several components, with surfactants among the most important. From both economic and environmental perspectives, the amount of surfactant should be minimized while still achieving the required performance. CMC gives a natural reference point for doing this.
In surfactant analysis, CMC is often discussed together with:
Effectiveness: how low a surface or interfacial tension value can be achieved with a surfactant system
Together, these parameters help compare different surfactants and find an optimal balance between performance, cost, and sustainability.
Further reading: Biosurfactant - Environmentally friendly alternative for many industries
A simple way to think about CMC is to consider how surfactant behavior changes with concentration.
Below the CMC: At very low surfactant concentrations, only a small number of molecules are present and the interface is still largely free, so surface tension changes only slightly. As the concentration increases, more surfactant adsorbs at the interface and surface tension decreases more rapidly.
Around the CMC: As concentration increases further, the interface becomes more and more occupied with surfactants. At some point, the interface becomes saturated with surfactant molecules and micelles start to form. This point is defined as the critical micelle concentration (Figure 2).
Above the CMC: Above the CMC, the addition of surfactants will no longer affect surface tension, as additional surfactant molecules mainly form micelles in the bulk solution instead of accumulating at the interface. These micelles often play a key role in solubilizing oils and hydrophobic components, which is important in detergency, emulsification, and many formulation challenges.
In practice, surface and interfacial tension measurements with tensiometers are the most widely used methods to measure CMC, because surfactants strongly affect interfacial behavior and the CMC often appears as a clear change in slope in the surface tension vs. concentration curve.
Critical micelle concentration can be defined by measuring surface tension as a function of surfactant concentration (Figure 2). The CMC point is identified as the point where the curve transitions from the steep region to the plateau.
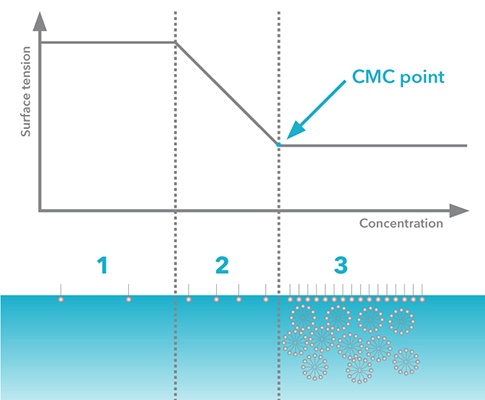
Figure 2. Surface tension as a function of surfactant concentration (logarithmic scale)
Surface and interfacial tension are typically measured using force tensiometers (with Wilhelmy plate or Du Noüy ring method) or optical tensiometers (pendant drop analysis). Force tensiometers are often the preferred solution for measuring CMC, because the concentration series and the measurement itself can be fully automated when the instrument is combined with dispensers. This makes it possible to change concentration stepwise, record surface tension at each step and locate the CMC point directly from the resulting curve, with minimal manual work.
Optical tensiometers can also be used to measure surface or interfacial tension over a range of concentrations, especially when sample volumes are limited or measurements need to be made above atmospheric pressure. In most cases, however, the concentration changes are made manually and the CMC point is identified from the data afterwards, rather than through a fully automated CMC workflow.
Other techniques can also be applied in specific systems:
Surface and interfacial tension are closely linked to how surfactants behave at interfaces, so changes around the CMC show up very clearly in their measurement data. Compared to many other techniques, surface tension methods are:
Direct – they probe the interfacial behavior that surfactants are designed to control.
Sensitive – even relatively small changes in adsorption can be detected.
Practical – they are well established, standardized and suitable for routine use in research, development and quality control.
CMC is relevant in many industrial and scientific contexts where surfactants play a central role. A few examples include:
Information about CMC helps formulators relate surfactant level to cleaning performance. By understanding when micelles start to form and how they influence solubilization of oils and soils, it becomes easier to balance performance, cost, and environmental impact in household and industrial cleaners.
In pharmaceutical and biotechnology formulations, surfactants are often added to stabilize proteins, peptides and colloidal drug carriers, or to improve wetting and dispersion of hydrophobic components. Because micelles form only above the critical micelle concentration, CMC data can help when assessing how a surfactant interacts with active ingredients and other excipients, and whether micelle formation is likely to play a role at the concentrations used.
In shampoos, body washes, facial cleansers, and similar products, CMC data can support choices around cleansing strength, foaming behavior, and mildness. Knowing how surfactants behave around and above the CMC can help formulators design products that are effective but still gentle to the user and the environment.
In areas such as wafer cleaning, coating of functional surfaces, or the preparation of battery electrodes and separators, surfactants are used to tune wetting and dispersion. CMC and related interfacial measurements can help engineers understand how surfactant concentration influences film formation, defect levels and process robustness.
Across these examples, CMC is one of the parameters that links molecular level surfactant behavior to practical performance, making it a useful reference point in both formulation and process development.
Critical micelle concentration is the surfactant concentration at which micelles begin to form in the bulk solution and additional surfactant mainly goes into micelles rather than to interfaces.
Below the CMC, surfactant molecules adsorb at interfaces and significantly reduce surface tension. As the system reaches and passes the CMC, the interface becomes saturated, and more of the added surfactant forms micelles in the bulk. Because of this, surface tension changes much less above the CMC.
For a specific surfactant under well‑defined conditions, you can agree on a reference value. However, CMC is not a fixed universal constant. Reported values depend on experimental method, sample history, and solution conditions, so they should always be interpreted together with this information.
Yes. Techniques such as surface tension, conductivity, and fluorescence measure different properties and respond differently to surfactant type, temperature, ionic strength, and additives. They may therefore yield somewhat different CMC values for the same system, even under otherwise similar conditions.
CMC helps compare surfactants, choose relevant concentration ranges, and understand how a system changes as surfactant is added. This supports formulation design, experimental planning, and the selection of appropriate analytical methods and instruments.
Surface tension measurement offers a practical and economical way to determine CMC and to characterize surfactant performance in real systems. Download our overview "Critical micelle concentration - What is it and how to measure it" below to learn more.

This article was first published April 3rd 2018, and has since been updated for completeness.
Surface tension is a property of a liquid surface: a net inward force that pulls the surface together and makes it act like a thin elastic film.
A wetting agent is a surface-active molecule used to reduce the surface tension of water.
The term surfactant comes from the word surface active agent. At the interface, they align themselves so that the hydrophobic part is in the air and the hydrophilic part is in water. This will cause a decrease in surface or interfacial tensions.
Surface tension plays an important role in Li-ion battery slurry optimization.
Surface tension plays an important role in the electroplating solution.
Anna Junnila is Customer Care Manager at Biolin Scientific. She takes pride in making advanced technology accessible for every user and is committed to guiding customers through every stage of their research journey. She holds an MSc in Electronics and Electrical engineering from Aalto University.
