A self-cleaning surface is any surface with the ability to readily remove any dirt or bacteria on it. This means that, for example, when raining, the water from the rain will clean the windows without a need for any external intervention. Self-cleaning surfaces can be divided into three different categories; superhydrophilic, photocatalytic and superhydrophobic.
Probably the simplest idea for self-cleaning is to make the surface superhydrophilic. On the superhydrophilic surface the water will spread on it completely forming a very thin layer of water. Generally, the term is used for surfaces with water contact angles below 5 degrees. Cleaning occurs when a thin sheet of water runs off the surface, washing away any loose dirt. One strategy for the fabrication of superhydrophilic surface is to combine hydrophilic surface chemistry with surface roughness.
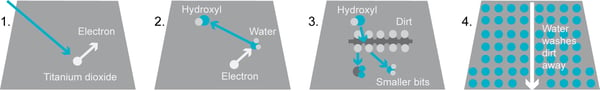
The first commercial self-cleaning windows were based on photocatalytic reaction. The glass is coated with thin titanium dioxide layer. UV illumination (1) makes the electrons of the TiO2 – surface to break water molecule into hydroxyl radicals (2). These radicals react with organic dirt, breaking them into smaller particles (3). The small particles are then easily washed away by water due to the superhydrophilic nature of the surface (4).
Superhydrophobic, self-cleaning surfaces are based on high water contact angles. A drop of water is almost spherical on the surface and readily rolls off taking the dirt away with it. Superhydrophobicity is achieved by combination hydrophobic surface chemistry and surface roughness. Surfaces with water contact angles above 150 degrees and low contact angle hysteresis are considered superhydrophobic.
To hear more about superhydrophobic surfaces and how they can be tailored for real-life applications, watch the webinar by Prof. Robin Ras.
Understand how superhydrophobic surfaces are defined, how they work, and how to measure them using contact angle methods. Explore key industrial applications—from self‑cleaning and anti‑icing to drag reduction—and what to consider when evaluating durability.
Single-cell trapping can be done with the help of superhydrophobic and superhydrophilic patterns.
Advancing and receding angles should be measured as the low contact angle hysteresis is also a requirement for superhydrophobicity.
There are three contact angle measurement methods for superhydrophobic surfaces; static, advancing/receding and roll-off angle.
Blood-repellent surfaces are needed in medical devices that come in contact with blood. The traditional approach has been the use of antithrombotic surface treatments However, these coatings are prone to eventually wear-off. Superhydrophobic surfaces have been proposed as an alternative solution.
With increasing understanding of the superhydrophobicity, the measurement methods to quantify the degree of hydrophobicity deserve some thought.
This video will explain two main methods for measuring dynamic contact angle.
Superhydrophobic surfaces were an instant hit in the scientific community when they were introduced over two decades ago
