Surface tension is a property of a liquid surface that makes it behave like a thin, elastic film pulling the liquid together. On a molecular level, it arises from cohesive forces between liquid molecules.
A simple way to see this is with a glass of water. Have you ever overfilled your glass of water so that the water level is slightly higher than the rim and still the water remains in the glass? This seemingly gravity-defying observation is an example of surface tension in action.
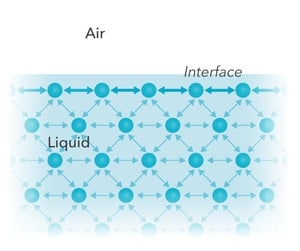
Imagine the interface between a liquid and a gas. Most of the molecules in the liquid bulk are surrounded by similar neighbors on all sides. The attractive forces between them are balanced in every direction, so the net force on a molecule in the bulk is essentially zero.
At the liquid surface, the situation is different. Some liquid molecules are at the interface, facing the gas phase. They have fewer neighbors on the gas side and experience a stronger pull towards the liquid bulk. This imbalance creates a net inward force and makes the surface behave as if it were covered by a "film".
This net inward pull on the molecules at the liquid surface, which effectively keeps the liquid together and tends to minimize the surface area, is what we call surface tension.
Mathematically, surface tension is defined as the force exhibited by the intermolecular forces divided by the length of the contact line between the phases. Therefore, the SI unit of surface tension is newton per meter (N/m).
Because the forces involved are very small, surface and interfacial tension are most often reported in millinewtons per meter (mN/m) Water, for example, has a relatively high surface tension of about 72 mN/m in room temperature due to its strong hydrogen bonds.
Many methods for measuring surface tension are based on directly measuring the force per unit length at the surface using a probe such as a ring, plate or rod. Other techniques use the shape of a droplet to calculate the tension from the balance of forces.
Modern tensiometers automate these principles and make it straightforward to follow how surface or interfacial tension changes with time, temperature or liquid composition.
If you want to learn about specific techniques, take a look at our article on 3 ways to measure surface tension.
For most pure liquids, surface tension decreases as temperature increases. Higher temperature increases molecular motion, which weakens the cohesive forces at the surface, lowering surface tension.
Surfactants are surface‑active molecules with a hydrophilic head and a hydrophobic tail. They adsorb at the surface and replace some of the liquid molecules there, weakening the cohesive forces. As a result, they lower surface tension and improve wetting, spreading and cleaning performance.
Surface tension usually refers to the tension at a liquid–gas interface, such as water–air. Interfacial tension refers to the tension at the interface between two immiscible liquids, such as oil and water.
Surface and interfacial tension affect wetting, spreading, foaming, emulsification and droplet formation. This makes them critical in applications such as coatings and paints, detergents and cleaning, inkjet printing, oil recovery, pharmaceuticals, food and cosmetics.
To learn more about surface tension and how it can be measured, download the white paper below.

Editor's note: This article was first published on Oct 18, 2018 and has since been updated for completeness.
Learn what critical micelle concentration (CMC) is, why it matters for surfactant performance and how it is commonly measured using surface tension measurements.
A wetting agent is a surface-active molecule used to reduce the surface tension of water.
The term surfactant comes from the word surface active agent. At the interface, they align themselves so that the hydrophobic part is in the air and the hydrophilic part is in water. This will cause a decrease in surface or interfacial tensions.
Surface tension plays an important role in Li-ion battery slurry optimization.
Surface tension plays an important role in the electroplating solution.
When measuring contact angles or making surface tension measurements with a pendant drop, selecting the correct tip or needle for your liquid is crucial.
The surface tension of water is about 72 mN/m at room temperature which is one of the highest surface tension for liquid.
Surface tension is a quantitative measure that can be correlated with a solution’s ability to remove dirt.
Anna Junnila is Customer Care Manager at Biolin Scientific. She takes pride in making advanced technology accessible for every user and is committed to guiding customers through every stage of their research journey. She holds an MSc in Electronics and Electrical engineering from Aalto University.
