
Surface cleanliness is one of the most important factors behind good adhesion and coating quality, and therefore an important topic in many industrial areas. Even if a surface looks clean to the eye, small amounts of contamination can dramatically reduce adhesion and lead to coating failure and poor printing quality. Contact angle measurements offer a fast and sensitive way to check whether your cleaning process is effective enough.
In this article, we look at how contact angle can be used to evaluate cleanliness across different types of surfaces, and how it can be applied in practical cleanliness control.
Contact angle measurement with water is a fast, quantitative way to verify whether a cleaning step has improved surface cleanliness.
For most engineering surfaces (metals, oxides, glass, many polymers), hydrophobic contamination increases water contact angle, while effective cleaning lowers it.
On polymers, especially very low‑energy ones such as fluoropolymers, dynamic (advancing/receding) contact angles often give a more reliable picture of cleanliness than static water contact angle alone.
Any organic or inorganic contamination between a surface and a coating, adhesive, or printed layer will weaken the interface. Even very thin contamination layers can:
Reduce wetting of coatings or adhesives
Cause poor coverage and local coating defects
Lower adhesion strength and increase the risk of delamination
Surface cleanliness is therefore critical in a wide range of applications, including metal coating and painting, polymer film printing and lamination, medical devices and diagnostics, as well as electronics and semiconductor manufacturing.
Because cleanliness is so important, it is essential to have a method that goes beyond visual inspection. Contact angle measurements provide a non‑destructive, quick and easy to standardize method for cleanliness evaluation.
A contact angle measurement quantifies how a liquid droplet (commonly water) wets a solid surface:
Cleaning changes the surface by removing low‑energy contaminants such as oils, residual processing additives, or adsorbed organic contaminants from the surface. As a result, the measured contact angle changes. In many practical cases:
This “cleaner = lower contact angle” rule is a good first approximation for many high‑ and medium‑energy engineering surfaces: metals and metal oxides, glass, and many polymers used in coating and bonding applications, where typical contamination is hydrophobic.
However, contact angle does not distinguish “clean vs dirty” by itself – it responds to whatever material is present at the surface. If residues from more polar materials remain after a cleaning step, for example from some solvents or detergents that have not fully evaporated or been rinsed away, they can also lower the water contact angle even though foreign material is still present. This is why cleanliness should always be defined relative to a well‑understood reference state for your own surface and process.
Clean metal surfaces have high surface free energies which can be seen as low water contact angles. On metals and metal oxides, contamination such as oils, greases, and organic films typically makes the surface more hydrophobic and increases the water contact angle. When the surface is properly cleaned, these contaminants are removed and the contact angle decreases.
In the table below, water contact angle on a copper and nickel surface is measured before any cleaning procedure and immediately after.1 Most of the contamination is already removed after ultrasonic cleaning in soap solution but contact angle can be further reduced with additional cleaning steps.
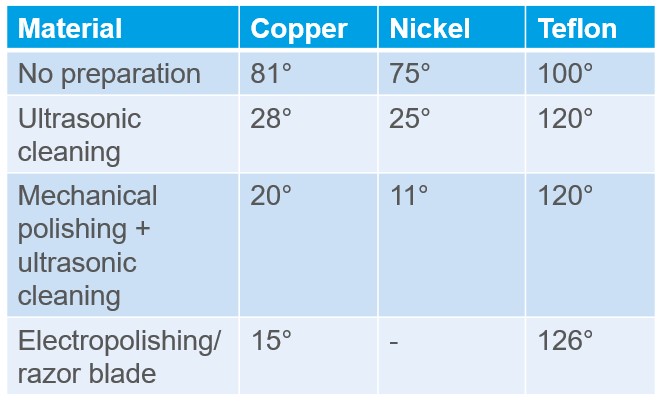
On copper and nickel, each more thorough cleaning step lowers the water contact angle, indicating a cleaner, higher‑energy surface and better conditions for adhesion. On Teflon, which is a very low‑energy fluoropolymer, the opposite trend appears: cleaning increases the water contact angle toward the intrinsic high value of the polymer. This is an important reminder that “cleaner = lower contact angle” does not always hold for fluoropolymers.
As the Teflon example above shows, polymers can behave differently from metals when it comes to cleanliness and contact angle.
In many practical applications, the contamination on polymers is hydrophobic and low‑energy (fingerprints, lubricants, residual siloxanes, mold‑release agents). For these situations:
Contamination usually increases the water contact angle and makes wetting and adhesion worse.
Cleaning or activation (plasma, UV/ozone, suitable solvents/detergents) removes low‑energy residues and often introduces polar groups, so the water contact angle decreases and wettability improves.
The important exception is very low‑energy polymers such as PTFE and some other fluoropolymers. Their clean, smooth surfaces already have intrinsically high water contact angles. If they pick up more polar material at the surface (for example surfactants or eco‑corona/biofilm), the water contact angle can actually decrease, and careful cleaning may raise it back towards the intrinsic high value. In the Teflon data above, the last preparation step with a razor blade increases the measured contact angle mainly because it adds roughness, not because the surface becomes chemically cleaner. This is why it is essential to define “clean” relative to a well‑understood reference state for each specific polymer.
In addition to a single static contact angle, it is often useful to look at advancing and receding contact angles, and the hysteresis between them. These so-called dynamic contact angle measurements are particularly informative on polymer surfaces, which are more prone than metal oxides to molecular rearrangement, additive migration, and low‑energy contamination. Such effects often show up much more clearly in the receding angle and hysteresis than in one static value, making dynamic measurements especially valuable for cleanliness and process control on polymers.
Once you understand how your surface behaves, you can use contact angle measurements as part of routine cleanliness or surface preparation quality control. The idea is always the same: to compare a well‑understood reference state with the cleaned or prepared state, and link the change in contact angle to real performance. A practical workflow would be:
During process development, measure contact angles on samples with known good and poor adhesion or coating quality. Define a target contact angle range (for metals often static water angle; for polymers including advancing/receding angles) that consistently corresponds to acceptable performance.
Standardize the probe liquid (often water), drop size, dosing speed, measurement time, and environmental conditions. For metals, a single water contact angle may be sufficient; for polymers, include dynamic angles and hysteresis.
Measure contact angle on a small number of representative samples per batch or shift. If values drift outside the defined range, you know the cleaning or surface preparation step needs attention before coating, bonding, or printing.
Automated contact angle instruments can make this process fast, repeatable and easy to document, without changing the basic principle: contact angle tells you whether cleanliness is where it needs to be for reliable adhesion and coating quality.
Surface inspection before coating or bonding
How to solve adhesion issues with contact angle measurements
Evaluate the influence of surface roughness on wettability through the Wenzel equation
Silicon wafer cleanliness evaluation through contact angle measurement
1. Which surfaces can I use contact angle on for cleanliness evaluation?
Contact angle can be used on many solid surfaces, including metals and metal oxides, polymers, coatings, glass and ceramics, and silicon wafers. The exact interpretation of the results depends on the material, but the general principle—comparing “before cleaning” and “after cleaning” states—remains the same.
2. Does a lower water contact angle always mean a cleaner surface?
Often, yes, especially for high‑ and medium‑energy surfaces where typical contamination is hydrophobic (oils, siloxanes, mold‑release agents). In those cases, contamination raises the contact angle and effective cleaning lowers it. However, for very low‑energy materials such as PTFE and some other fluoropolymers, a carefully cleaned surface can actually have a higher contact angle than one that has picked up more polar residues. That is why it is important to define cleanliness relative to a well‑understood reference state for your own material.
3. Why are advancing and receding contact angles useful for cleanliness evaluation on polymers?
Dynamic contact angle measurements reveal both the overall wettability and the uniformity of the surface. The advancing angle shows how hydrophobic or hydrophilic the surface is, while the receding angle and the hysteresis between them are very sensitive to residual contamination as well as chemical and topographical inhomogeneity. This makes them powerful tools for detecting when a polymer surface is not as clean or consistent as it should be.
4. How do I turn contact angle into a practical pass/fail cleanliness test?
The key is to link contact angle ranges to real performance on your own components. During process development, measure contact angles on surfaces known to give good and poor adhesion or coating quality, and identify contact angle ranges that reliably distinguish them. These ranges then become your practical criteria. In production, if measured contact angles fall outside the acceptable window, you know the cleaning or surface preparation step needs to be adjusted before proceeding.
Contact angle measurements offer a practical way to move beyond visual inspection to quantify surface cleanliness. By comparing contact angle before and after cleaning, and linking those values to actual coating or adhesion performance, you can turn cleanliness into a controlled, measurable parameter instead of a guess. The same principles apply across different materials, from metals and oxides to polymers and wafers, as long as cleanliness is defined relative to a well‑understood reference state.
If you would like to learn more about using contact angle measurements to predict adhesion, please watch our free webinar below.

Editor's note: This post was originally published April 12th, 2022 and has since been updated for completeness.
References:
1. Horsthemke and J.J. Schröder, “The wettability of industrial surface: contact angle measurements and thermodynamic analysis”, Chemical Engineering and Processing: Process Intensification 19 (1985) 277Learn why paint and ink fail to stick. A quick guide to understanding wettability and adhesion for better product performance.
Standard contact angle measurement considers the surface's chemical properties. The influence of surface roughness is added by utilizing the Wenzel equation.
Cohesion and adhesion are fundamental concepts in the study of physics and chemistry, playing crucial roles in various natural and industrial processes.
Surface inspection is done to guarantee optimum surface properties for coating and bonding
A spreading coefficient is a measure of the wetting behavior of a liquid on a solid surface.
Wetting and adhesion analysis has been added to our all-inclusive OneAttension software and is available for download for all OneAttension customers.
Depending on the strength of these forces, the adhesion failure can be either adhesive, cohesive, or substrate failure.
One of the main challenges in PCB manufacturing is the adhesion of the conformal coating. Contact angle measurements can be used to predict adhesion.
Anna Junnila is Customer Care Manager at Biolin Scientific. She takes pride in making advanced technology accessible for every user and is committed to guiding customers through every stage of their research journey. She holds an MSc in Electronics and Electrical engineering from Aalto University.
