
Wettability has been recognized as one of the most important factors affecting enhanced oil recovery (EOR). Several studies have shown the influence of different EOR agents on the reservoir rock wettability and oil recovery rates. However, most of these studies are not considering the reservoir conditions i.e. the high temperature and high pressure.
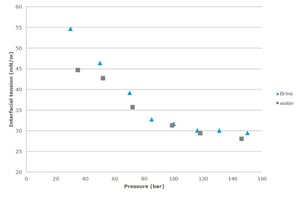
Low interfacial tension (IFT) between the crude oil and the injected fluid is generally considered to be an important requirement for mobilization and displacement of the oil. Using CO2 as an injection fluid is gaining a lot of interest since there is also a possibility for simultaneous CO2 capture and storage. To be able to predict the CO2 flooding capacity, it is necessary to study the interfacial tension change between CO2 – water / oil with pressure, temperature and CO2 concentration. The influence of pressure on interfacial tension between CO2 – water/brine and CO2-crude oil has been studied. Generally, it has been found that the IFT decreases with increasing pressure for both systems. This has been linked to adsorption of CO2 molecules and the higher CO2 solubility at elevated pressures.
Surfactant flooding is another much used EOR method. The two main functions of surfactants in enhanced oil recovery is either to reduce the interfacial tension or wettability alteration. For wettability alteration with surfactants two different mechanisms have been proposed; coating and cleaning mechanisms. In coating mechanism, the surfactant is adsorbed onto the oil-wet solid surface. Cleaning mechanism, on the other hand, refers to a process where surfactant molecules complex with contaminant molecules adsorbed from crude oil to strip them off the rock surfaces.
It is well-known that the surfactant behavior can change drastically at high temperatures. The effect of pressure is less studied. However, to be able to measure water-based surfactants near and above 100 °C, high pressures are needed to prevent boiling.
If you would like to see more examples how pressure affect the wettability, please download the slide deck below.
Smart water flooding in EOR: how tailored brine composition changes reservoir wettability in sandstone and carbonate formations to improve oil recovery.
Most commonly used methods to study reservoir wettability are Amott-Harvey, USBM, and sessile drop contact angle.
In enhanced oil recovery, wettability plays an important role as it determines the interactions between the solid (rock) and the liquids in the reservoirs (crude oil, brine). Wettability has been recognized as one of the key parameters controlling the remaining oil-in-place.
Nanoparticles alone or integrated with conventional enhanced recovery processes have shown promising performance in improving oil recovery.
There are three commonly used wettability measurement techniques for oil reservoir characterization; Contact angle, Amott-Harvey, and USBM.
Carbonate reservoirs are characterized as intermediate to oil- wet. Altering the wettability of the carbonates has been proposed as one of the main mechanisms for enhanced oil recovery.
Different enhanced oil recovery methods are used to alter the wettability of the reservoir rock. To study the wettability alteration at the reservoir conditions, an instrument where the measurements can be done at high pressures and temperatures are needed.
Unconventional oils, such as heavy oil, extra heavy oil, and bitumen, normally exist tightly on host solids such as rocks, sands and clay minerals. Successful liberation of unconventional oil from solids is essential for effective recovery.
