
Surface tension measurements are important in many industrial applications. There are several ways to measure surface tension such as Du Noüy ring, Wilhelmy plate, and pendant drop. The first two are force-based methods whereas the pendant drop is done with the optical tensiometer.
Surface and interfacial tension measurements can be performed optically using pendant drop shape analysis. The shape of the drop hanging from a needle is determined from the balance of forces which include the surface tension of the liquid being investigated.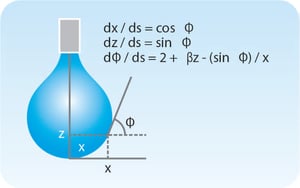 The surface or interfacial tension can be related to the drop shape by the equation;
The surface or interfacial tension can be related to the drop shape by the equation;
γ = ΔρgR0/β
where γ is the surface tension, Δρ is the density difference between fluids, g is the gravitational constant, R0 is the drop radius of curvature at the apex and β is the shape factor. β can be defined through the Young-Laplace equation expressed as 3 dimensionless first-order equations as shown in the image.
Modern computational methods using iterative approximations allow for solutions of the Young-Laplace equation to be found. Thus, the surface or interfacial tension between any two immiscible fluids with known densities can be determined. For optical tensiometry the size of the droplet is important, and it should have a tear or pendant shape. Care must be taken to ensure that the needle tip is not influencing the drop shape. When measuring surface tension, the density difference between liquid and gas (usually air) is large enough that volumes between 5 μl to 20 μl are generally sufficient to provide pendant or tear shapes. When measuring interfacial tensions, both density difference and interfacial tension affect the required volume to achieve a pendant or tear shape drop. As a guideline, the smaller the density difference, the larger the volume required.
The force-based methods are somewhat more established, and several standards describe the surface tension measurements with the Du Noüy ring method. The pendant drop method is however gaining popularity due to various benefits the method can offer.
1. Small sample volumes
The pendant drop method is especially suited when the sample volumes available are small. With the force-based method, the minimum sample volume is few milliliters at best but with the pendant drop, about 100 μl or even less is sufficient to conduct the measurement. This is important for example when measuring biological samples that are typically limited in volume. Small sample volumes are also beneficial when hazardous samples are measured.
2. Easy liquid handling
The pendant drop method can be done by using the disposable pipette tip. Sample can be easily drawn to the pipette. After the measurements, the pipette tip can be changed. This reduces the risk of having contaminants in the sample. It also makes the measurement of sticky samples (such as paints and glues) easier and faster, as there is no need to clean the measurements cup and probes after the measurement is completed.
3. Probe quality doesn’t affect the results
The force-based methods utilize the measurement probe i.e. the Du Noüy ring or the Wilhelmy plate to conduct the measurements. The dimensions of the probe are critical for successful surface and interfacial tension measurements. The probes are somewhat fragile and need to be handled with care to ensure the correct shape and size of the probe. With the pendant drop method, this is not an issue as the method is based on the free-hanging drop shape.
4. Check the purity of liquids before contact angle measurements
Pendant drop measurement can also be combined with the contact angle measurements easily. It is thus possible to check the purity of the liquid used before contact angle measurements. In the OneAttension software, it is possible to determine the acceptable surface tension range for the liquid used. When the contact angle measurement is conducted the software will automatically check the purity of the liquid before droplet deposition.
A practical guide to choosing a force tensiometer: what to look for in usability, software licensing, maintenance and long-term value — beyond the spec sheet.
Surface tension is a property of a liquid surface: a net inward force that pulls the surface together and makes it act like a thin elastic film.
Learn what critical micelle concentration (CMC) is, why it matters for surfactant performance and how it is commonly measured using surface tension measurements.
A wetting agent is a surface-active molecule used to reduce the surface tension of water.
The term surfactant comes from the word surface active agent. At the interface, they align themselves so that the hydrophobic part is in the air and the hydrophilic part is in water. This will cause a decrease in surface or interfacial tensions.
Surface tension plays an important role in Li-ion battery slurry optimization.
Surface tension plays an important role in the electroplating solution.
When measuring contact angles or making surface tension measurements with a pendant drop, selecting the correct tip or needle for your liquid is crucial.
The surface tension of water is about 72 mN/m at room temperature which is one of the highest surface tension for liquid.
