Adhesion is the attraction between two dissimilar phases. There is no single theory to explain adhesion, but it is commonly divided into mechanical interlocking and physical and chemical bonding. Most typically adhesion occurs because of the combination of different mechanisms explained below.
Mechanical interlocking happens when two dissimilar phases attach to one another by mechanical forces only. This typically requires a somewhat rough surface where the other material can penetrate. 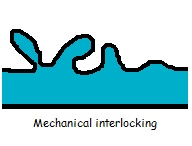 For example, a low viscosity adhesive that flows into pores and cracks in the surface of a material. This results in mechanical anchoring between two components when the adhesive has hardened. With this knowledge, you can assume that rough surfaces would be easy to fix together with glue. However, a too rough surface can lead to stress peaks and insufficient filling of the surface structures. How well the adhesive fills the cavities on the surface, depends on the shape of the cavities as well as the viscosity and the surface tension of the adhesive.
For example, a low viscosity adhesive that flows into pores and cracks in the surface of a material. This results in mechanical anchoring between two components when the adhesive has hardened. With this knowledge, you can assume that rough surfaces would be easy to fix together with glue. However, a too rough surface can lead to stress peaks and insufficient filling of the surface structures. How well the adhesive fills the cavities on the surface, depends on the shape of the cavities as well as the viscosity and the surface tension of the adhesive.
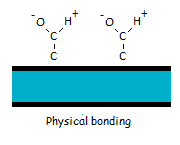
Physical bonding consists of van der Waals forces and is always present. The strength of the van der Waals forces is rather weak and they usually don’t contribute that much on the total bonding strength.
Chemical bonding includes covalent, ionic and metallic bonding which are much stronger than physical bonds. 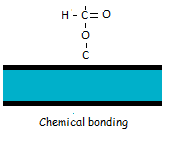 Chemical bondings are responsible for cohesive forces inside the material itself which can be very strong. However, chemical bonding between two dissimilar materials is much more complicated and there are typically only a few available bonding sites. One of the most used methods to increase the number of bonding sites is plasma treatment.
Chemical bondings are responsible for cohesive forces inside the material itself which can be very strong. However, chemical bonding between two dissimilar materials is much more complicated and there are typically only a few available bonding sites. One of the most used methods to increase the number of bonding sites is plasma treatment.
Based on the above mention bonding mechanisms, it is evident that the coating should spread on the substrate to achieve good adhesion. Good spreading will facilitate the penetration of the coating to surface structures as well as an introduction to new binding sites. Wettability of the substrate is thus often evaluated. As contact angle is a measure of wettability, it is used to evaluate bonding issues.
To read more about contact angle and how it can be utilized, read the overview below.
Learn why paint and ink fail to stick. A quick guide to understanding wettability and adhesion for better product performance.
Learn how to use contact angle measurements to evaluate surface cleanliness on different materials and link cleaning steps to reliable adhesion and coating quality.
Standard contact angle measurement considers the surface's chemical properties. The influence of surface roughness is added by utilizing the Wenzel equation.
Cohesion and adhesion are fundamental concepts in the study of physics and chemistry, playing crucial roles in various natural and industrial processes.
Surface inspection is done to guarantee optimum surface properties for coating and bonding
A spreading coefficient is a measure of the wetting behavior of a liquid on a solid surface.
Wetting and adhesion analysis has been added to our all-inclusive OneAttension software and is available for download for all OneAttension customers.
Depending on the strength of these forces, the adhesion failure can be either adhesive, cohesive, or substrate failure.
One of the main challenges in PCB manufacturing is the adhesion of the conformal coating. Contact angle measurements can be used to predict adhesion.
