
Crosslinking and collapse of surface‑adhering layers can be measured with QSense QCM‑D by monitoring changes in resonance frequency (Δf) and dissipation (ΔD) as the hydrated layer releases solvent and transforms into a thinner, more compact state. Solvent release and layer collapse typically appear as an apparent loss of coupled mass (Δf↑) and a decrease in dissipation (ΔD↓) as the film becomes less hydrated and more rigid. By combining these QCM-D signals with modelling, you can quantify changes in thickness and mechanical properties, and infer relative changes in hydration during crosslinking and collapse of for example polymer brushes, multilayers, hydrogels and other interfacial films.
Surface‑bound layers such as polymer brushes, polyelectrolytes, hydrogels and biomolecular films are widely used to tailor interfacial properties – for example to control wetting, protein adsorption, or cell and bacteria adhesion. Their behaviour is strongly influenced by how hydrated they are at the surface, and by whether the layer is in an open, swollen state or in a more compact, crosslinked state. In this post, we show how crosslinking and collapse of such interfacial films can be characterized with QSense QCM‑D.
Polymeric and biomolecular layers are used in many applications, including biomaterials, antifouling and antimicrobial coatings, sensors, filtration membranes, and cleaning and hygiene products. By adjusting the structure and hydration of the interfacial layer, you can influence, for example, wetting, protein adsorption, cell and bacteria adhesion, antibacterial performance and even drug delivery or other stimulus‑responsive behaviours.
To design layers with the desired performance, it is important to characterize and understand their conformational behaviour at the interface, including the degree of hydration and transitions between hydrated, collapsed and crosslinked states (Fig. 1).
QSense QCM‑D detects changes in the total load, i.e. mass, coupled to the sensor and in the energy dissipation. Together, these signals reveal solvent uptake and release, as well as changes in the structure and mechanics of the interfacial layer, making it possible to follow crosslinking and collapse in real time.
Figure 1. Schematic illustration of a hydrated, thick interfacial film (left side) that releases water and collapses into a thinner layer at the surface (right side).
QSense QCM‑D is an acoustic technique that measures how a quartz sensor’s resonance frequency changes when material and liquid are coupled to its surface. Because the oscillation “feels” both the film and the liquid that moves with it, QCM‑D is sensitive to the so‑called hydrated mass of the interfacial layer – the dry material plus its dynamically coupled water.
The swelling and collapse of polymer and biomolecular films can therefore be characterized with QSense QCM‑D by following how this hydrated mass and the film mechanics change over time. This makes it well suited to monitor:
When a hydrated interfacial layer (for example a polymer, polysaccharide or protein film) collapses or becomes crosslinked, QSense QCM‑D typically records the following:
Frequency shift (Δf):
Δf↑ (frequency increases) → decrease in the total hydrated mass coupled to the sensor as part of the previously coupled water is released and the layer contracts.
Dissipation shift (ΔD):
ΔD↓ (dissipation decreases) → the layer becomes less hydrated, more compact and mechanically stiffer, so energy losses per oscillation are reduced.
In contrast, when the layer swells and takes up solvent:
Δf↓ → increase in hydrated mass due to both the film and dynamically coupled solvent.
By following Δf and ΔD at multiple harmonics and combining the data with viscoelastic modelling, you can extract:
As an example, let’s look at the transitions between hydrated and dehydrated states of polymer brushes made of chitosan. At low pH, the chitosan brush is highly protonated and in a hydrated, swollen state, while at higher pH it becomes less charged and collapses into a more dehydrated, compact layer. In addition, suitable anions can be used to induce crosslinking within the brush.
The results, Fig. 2, show how the thickness of the chitosan brush varies as it undergoes swelling and collapse when pH and counter‑anion type are changed:
These measurements reveal how chitosan brush conformation is controlled by pH and counter‑ion choice, and how crosslinking can be used to lock an interfacial polymer layer into a more collapsed state.
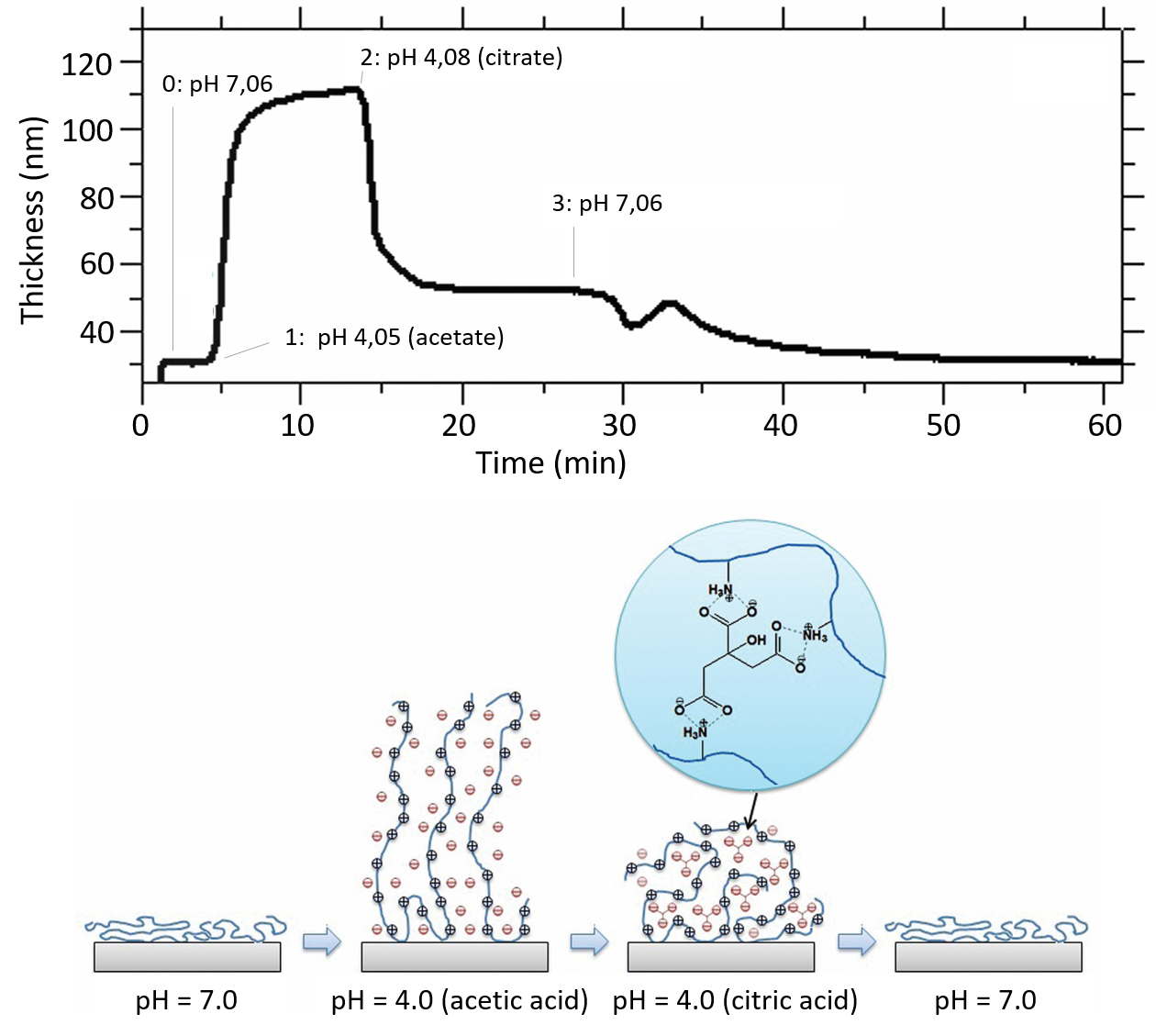
Figure 21. (Top) The thickness of the Chitosan brush layers when exposed to different solution pH and counter-ions, , showing swelling, collapse and anion‑induced crosslinking. (Bottom) Schematic illustration of the structure of the chitosan brush layer as a function of pH and counter-ion type.
The following generic protocol can be adapted to study transitions in polymer, biopolymer or biomolecular films (e.g., brushes, multilayers, hydrogels, protein layers).
1. Prepare the interfacial layer
2. Establish a hydrated reference state
3. Induce collapse or dehydration
4. Introduce crosslinking conditions
5. Analyse and compare states
Understanding and controlling interfacial layer conformation is relevant in many areas, for example:
In all these contexts, QSense QCM‑D helps you link molecular‑scale conformational changes (hydrated vs. collapsed vs. crosslinked) to macroscopic performance.
Interfacial films such as polymer brushes, multilayers, hydrogels and biomolecular coatings are more or less hydrated and viscoelastic depending on their molecular conformation at the surface. This conformation has a major impact on interfacial properties and on interactions with the surrounding environment, for example protein adsorption, cell or bacteria adhesion. QSense QCM‑D enables straightforward characterization of these conformational changes. By following mass (Δf) and dissipation (ΔD) in real time, you can detect transitions between more hydrated and more collapsed or crosslinked states, quantify changes in thickness and mechanical properties, and evaluate how pH, ion type and other conditions influence interfacial layer structure and apparent hydration.
Download the overview to read more about what information you can obtain with QSense QCM‑D and how it can be used to study swelling, adsorption/desorption, and other interfacial phenomena.

1. H-S Lee, et al., J. Mater. Chem., 22, 19605, 2012
Editor’s note: This post was originally written and published in 2018 by a former Biolin Scientific colleague. It has been updated for clarity and comprehensiveness while keeping the original work as the foundation.
1. Figures are reproduced from Ref. 1 with permission from The Royal Society of Chemistry
QSense QCM-D reveals lipid bilayer formation and hydrated model membrane structures that can be difficult to capture with other methods.
QSense QCM-D reveals in real time how thin films degrade and are removed, from cleaning and detergency to corrosion, etching and degradable coatings.
Learn how QSense QCM‑D reveals time‑resolved reagent–mineral interactions, linking surface chemistry to flotation performance in mineral processing.
Learn how QSense QCM-D reveals protein–surface interactions and adds interface-focused insight to biopharmaceutical formulation and stability work
Learn how QSense sensors enable application‑relevant biointerface interaction analysis and explore our sensor offering for different areas
Learn how QSense QCM D can be used to analyze swelling of thin films, including magnitude and dynamics.
Read about how molecule-surface interaction processes such as adsorption and desorption can be analyzed with QCM-D.
Learn best practices and step-by-step methods for accurate QCM-D coating thickness measurement on QSense sensors using QSense Omni.
Compared to QCM, QCM-D measures an additional parameter, and provides more information about the system under study.
Discover how QCM-D analysis reveals real-time etching dynamics, helping optimize cleaning processes and protect surfaces from unwanted damage.
