
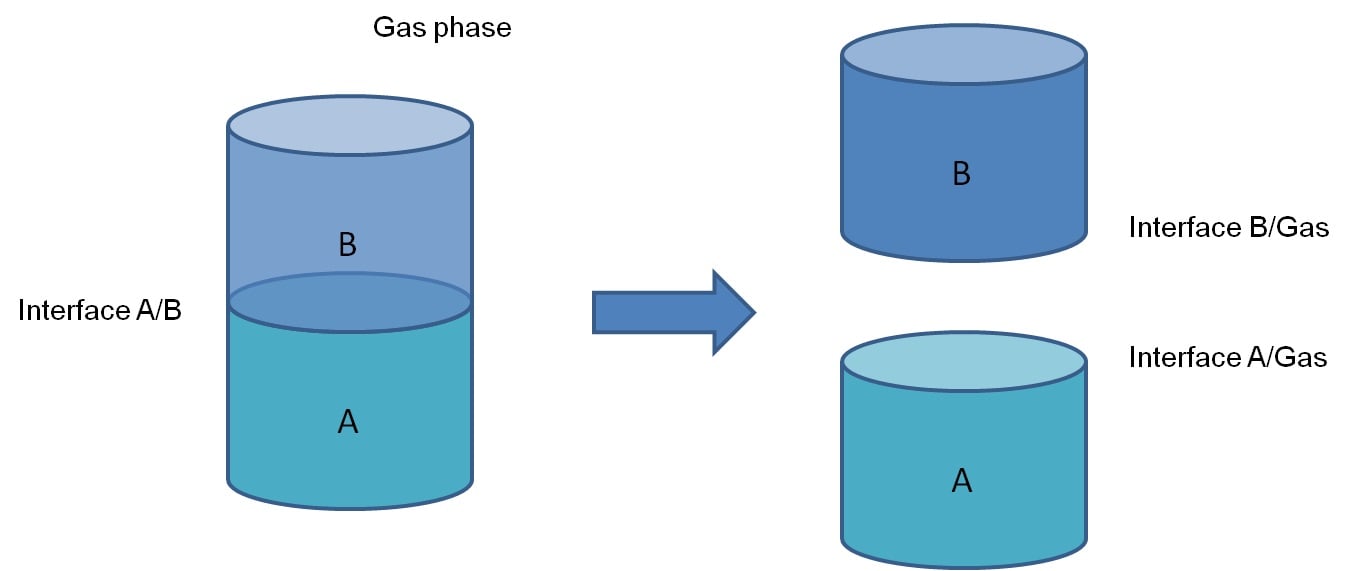
Work of adhesion is defined as a work required to separate two phases from each other. When the two phases are in contact with each other, there is an interface between them with a certain interfacial energy, γAB. When the two phases are separated, this interface will disappear but two new are formed. These are the interface between phase A and air (γA) and the interface between phase B and air (γB). The work that is required to separate these is work of adhesion and described by Dupré as: WAB = γA + γB – γAB.
Now, if the two phases are solid and liquid, the equation can be written as
WSL = γS + γL – γSL
,where γS is the surface free energy of the solid, γL is the surface tension of a liquid and γSL is the interfacial tension between solid and the liquid.
While the surface tension of the liquid is easy to measure, the other two are more cumbersome. To simplify the measurement of work of adhesion, the Dupré equation can be combined with the Young equation; γS = γSL + γLcosθY
which then gives Young-Dupré equation
WSL = γL(cosθ + 1).
This equation makes it possible to easily access the work of adhesion with simple contact angle measurements.
For example, work of adhesion between water drop and a glass slide would be measured by measuring the contact angle of water on the glass surface. For surface tension of water, you can use a literature value or measure it. With this information, the work of adhesion can be calculated.
To read more about contact angle measurements, please download the white paper through the link below.
Learn why paint and ink fail to stick. A quick guide to understanding wettability and adhesion for better product performance.
Learn how to use contact angle measurements to evaluate surface cleanliness on different materials and link cleaning steps to reliable adhesion and coating quality.
Standard contact angle measurement considers the surface's chemical properties. The influence of surface roughness is added by utilizing the Wenzel equation.
Cohesion and adhesion are fundamental concepts in the study of physics and chemistry, playing crucial roles in various natural and industrial processes.
Surface inspection is done to guarantee optimum surface properties for coating and bonding
A spreading coefficient is a measure of the wetting behavior of a liquid on a solid surface.
Wetting and adhesion analysis has been added to our all-inclusive OneAttension software and is available for download for all OneAttension customers.
Depending on the strength of these forces, the adhesion failure can be either adhesive, cohesive, or substrate failure.
One of the main challenges in PCB manufacturing is the adhesion of the conformal coating. Contact angle measurements can be used to predict adhesion.
