QSense QCM-D can be used to characterize model membranes and other lipid-based structures at surfaces by monitoring changes in resonance frequency, Δf, and dissipation, ΔD, in real time. These signals make it possible to follow lipid adsorption, vesicle rupture, supported lipid bilayer formation, and interactions between lipid-based structures and membrane-active molecules.
Because QCM-D measures the total coupled mass at the interface, including associated solvent, it is especially well suited for studying soft and highly hydrated lipid systems such as vesicles, supported lipid bilayers, and other membrane-like assemblies. This makes it possible not only to detect how much material is present at the surface, but also to distinguish between different lipid structures and follow configurational changes as they occur.
Model membranes are widely used in life science and materials research to study biological interfaces under controlled conditions. They provide simplified systems that mimic key features of cell membranes while allowing experiments to be performed with well-defined lipid compositions, surface chemistries, and environmental conditions.
These systems are important in many areas, including biosensor development, biomaterials, membrane-protein research, antimicrobial studies, and drug delivery. They are also used to investigate how molecules such as proteins, peptides, polymers, nanoparticles, surfactants, and drug candidates interact with lipid membranes.
To understand these systems, it is important to characterize not only whether lipids adsorb to a surface, but also how they are organized once they are there. For example, lipids may remain as intact vesicles, rupture and reorganize into a supported lipid bilayer, or undergo structural changes when exposed to external stimuli. Because these are interfacial and highly dynamic processes, they are well suited for study with QSense QCM-D.
QSense QCM-D is an acoustic, surface-sensitive, and label-free technique that provides real-time information about changes in mass andviscoelastic properties at the sensor surface. In membrane studies, this enables monitoring of both membrane formation and membrane interaction processes.
QCM-D has a long track record in model membrane research, with published studies of lipid vesicle adsorption and supported lipid bilayer formation dating back to 1998.1
One of the key advantages of QCM-D is that it measures the so-called hydrated mass. This means that the signal includes not only the lipid molecules themselves, but also the liquid dynamically coupled to the structure at the surface. This is particularly important for soft and hydrated lipid systems.
For example, intact adsorbed vesicles contain and couple a relatively large amount of solvent, whereas a planar supported lipid bilayer contains much less coupled solvent. Even if the amount of lipid material at the surface is similar, the QCM-D response is very different. This allows QCM-D to distinguish between lipid structures that would otherwise be difficult to differentiate.
During a QCM-D experiment, two main signals are recorded.
Frequency shift, Δf: The frequency shift reflects changes in the total mass coupled to the surface. In membrane studies, a decrease in frequency generally indicates adsorption or increased coupled mass, while an increase in frequency can indicate loss of material or restructuring into a less hydrated state.
Dissipation shift, ΔD: The dissipation shift reflects the viscoelastic properties of the surface-bound layer. A high dissipation signal typically indicates a soft, highly hydrated, and more deformable structure, such as a layer of intact vesicles. A lower dissipation signal is consistent with a more rigid and compact layer, such as a supported lipid bilayer.
Together, Δf and ΔD provide insight into:
A common application of QCM-D is the formation of a supported lipid bilayer by adsorption and rupture of lipid vesicles on a solid surface.
At the start of the experiment, schematically illustrated in Fig. 1, vesicles are introduced to the sensor surface. As they adsorb, the coupled mass increases because intact vesicles contain a large amount of associated water. This typically leads to a pronounced decrease in frequency and a relatively high dissipation signal.
As surface coverage increases, the vesicles may begin to rupture and fuse. During this structural transition, the QCM-D response changes significantly because the system reorganizes from a soft, highly hydrated vesicle layer to a thinner and less hydrated bilayer.
Once a supported lipid bilayer has formed, the final signal is characterized by a smaller mass load and much lower dissipation than the intact-vesicle layer. In this way, QCM-D makes it possible to follow the entire bilayer formation process in real time and distinguish intermediate stages of the transformation.
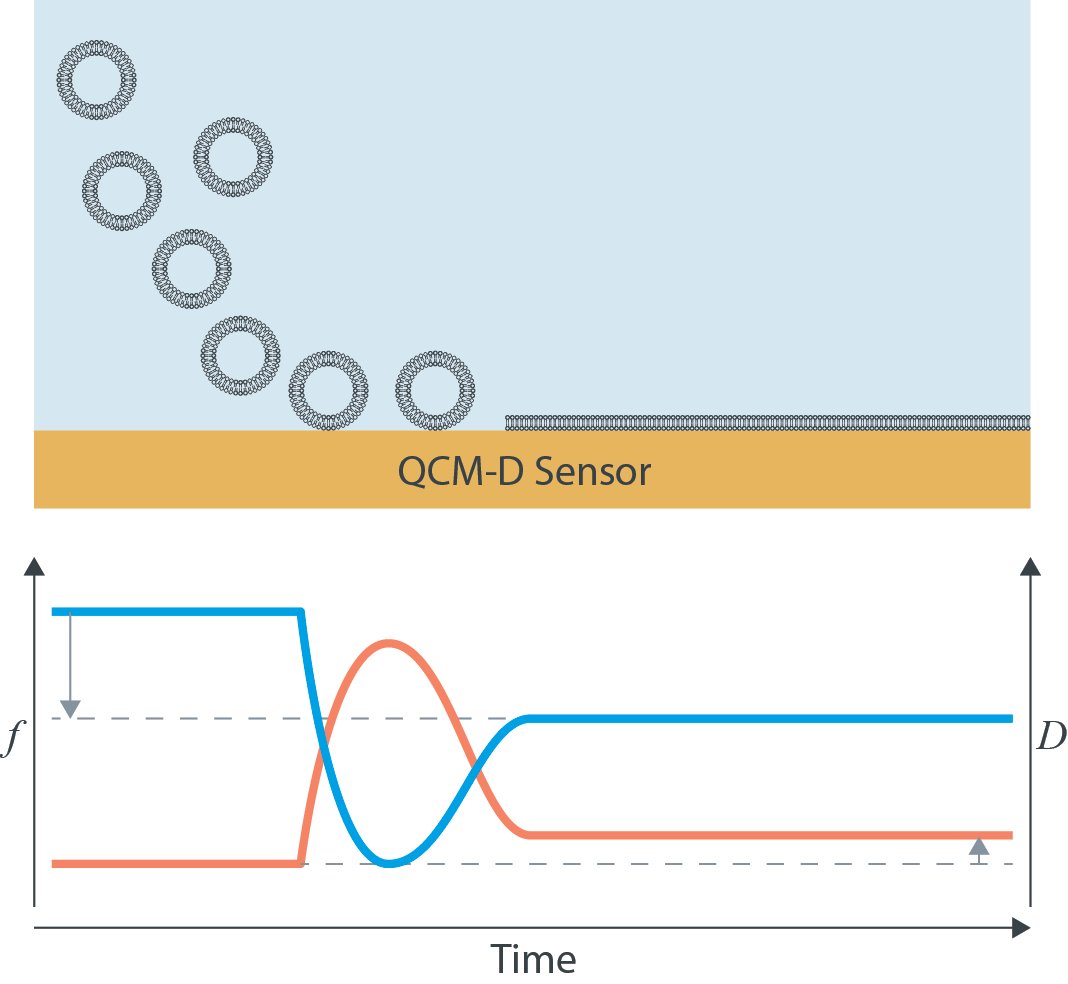
Figure 1. Schematic illustration of supported lipid bilayer formation from adsorbed lipid vesicles, monitored with QCM-D.
QCM-D is also widely used to study how other molecules interact with lipid-based structures. Once a stable model membrane has been formed, the system can be exposed to proteins, peptides, nanoparticles, surfactants, polymers, or drug candidates.
Depending on the interaction, QCM-D may reveal:
This makes QCM-D useful not only for forming model membranes, but also for studying membrane-active compounds and interfacial mechanisms relevant to biology, medicine, and materials science.
A generic workflow for studying model membranes with QCM-D can include the following steps.
1. Prepare the sensor surface: Choose a sensor surface suitable for the membrane system of interest, for example silica-coated sensors for supported lipid bilayer formation from vesicles. Clean and handle the sensors carefully to ensure reproducible results.
2. Establish a stable baseline: Equilibrate the sensor in the chosen buffer until stable frequency and dissipation baselines are obtained.
3. Introduce the lipid system: Inject the vesicle suspension or other lipid formulation and monitor the QCM-D response in real time as adsorption occurs.
4. Follow structural transitions: Observe whether the lipid structures remain intact, reorganize, rupture, or fuse into a more compact membrane layer. Use the combined Δf and ΔD response to interpret the structural state of the lipid layer.
5. Rinse and stabilize the formed layer: Rinse with buffer to remove loosely bound material and evaluate the stability of the membrane structure remaining at the surface.
6. Introduce interaction partners if relevant: If the aim is to study membrane interactions, introduce the molecule of interest after membrane formation and monitor the resulting response.
Model membranes are relevant in many research and development areas.
Biosensors and surface functionalization: Lipid layers can be used as biologically relevant coatings and recognition environments in biosensor development.
Biomaterials and biointerfaces: Model membranes help evaluate how materials interact with cell-membrane-like surfaces, which is important in implants and other biomedical applications.
Drug delivery and pharmaceutical research: Lipid systems are relevant for understanding membrane permeability, drug-membrane interactions, and delivery vehicle behavior.
Membrane-active molecules: QCM-D can be used to study the effects of peptides, proteins, detergents, nanoparticles, and other compounds on membrane structure and stability.
Fundamental membrane research: Supported lipid bilayers and vesicle systems offer controlled model platforms for studying interfacial membrane processes.
Ignoring hydration effects: The coupled solvent is a major part of the QCM-D response for lipid systems. This is not a complication, but an important source of structural information.
Overinterpreting a single overtone: Use several overtones to assess the consistency of the response and to better understand the viscoelastic behavior of the layer.
Not controlling buffer and surface conditions: Bilayer formation and membrane interactions are highly sensitive to buffer composition, ionic strength, pH, temperature, and surface chemistry. These parameters should be carefully controlled and documented.
QSense QCM-D provides a straightforward and highly informative way to characterize model membranes and other lipid-based structures at surfaces. By measuring changes in frequency, Δf, and dissipation, ΔD, in real time, it becomes possible to distinguish between vesicles, bilayers, and other membrane structures, and to follow their formation and transformation as they happen.
Because QCM-D is sensitive to hydrated mass as well as layer mechanics, it is particularly well suited for revealing structural and configurational changes in soft lipid systems. This makes the technique valuable for membrane research, biosensor development, biomaterials, and drug delivery applications.
Download the overview to read more about model membrane characterization with QSense QCM-D.
Editor’s note: This post was originally published in 2019 and has been updated for clarity, completeness, and broader application relevance while keeping the original content as the foundation.
Discover how QCM-D enables real-time, label-free analysis of supported lipid membrane formation, structure, and dynamics for advanced research
QCM-D was used to compare the potency and mechanisms of action of two different detergents in disrupting lipid membranes
Read about Prof. Jackman's experience using QCM-D to study surfactant-interaction with model membranes
Read about Prof. Jackman's experience using QCM-D in the field of membrane biophysics.
Watch the webinar to learn more about how to combine QCM-D and Neutron reflectrometry to examine membrane biochemistry at the solid-liquid Interface
